Sodium Silicate is an organic and chemical compound that is widely used in the chemical industry. This alkaline substance, also known as water glass, is obtained from the combination of sodium oxide and silica. According to the book “Chemical Technology Guide,” sodium silicate was first discovered by Van Helmont in 1640. This material is stable in neutral and alkaline solutions and is divided into two physical forms: solid and liquid.
In addition, sodium silicate takes on various forms (orthosilicate, metasilicate, disilicate, tetrasilicate) depending on the acid from which it is derived. Each of these forms has a specific application in the industry, making this substance one of the most important silicates in the industry. Therefore, sodium silicate exports are of great importance and profitability.
Soluble silicates of alkaline metals such as sodium and potassium were discovered by European chemists in the 1500s. However, it was not clear how this process was completed until the first industrial applications of sodium silicate were used in the mid-1800s in Europe and the United States in 1818. This discovery was made by Johann Nepomuk von Fuchs, a mineralogy professor. Alkaline soluble silicates, which are currently one of the largest volume of chemical compound productions in Western Europe, have actually been known since ancient times. Various types of soluble silicates were identified by some chemists in the 17th century and were put to use.
As sodium silicate has many applications in the chemical industry, Raag Trading Company is dedicated to supplying and exporting the highest quality sodium silicate. This product, which is manufactured based on international standards, meets the needs of domestic and foreign industrialists. In addition, Raag Trading Company offers sodium silicate at a lower price than other trading sites and with a lower profit margin in order to develop industries and meet the needs of domestic and foreign customers.
You can easily order this product through RAAG’s website and have it delivered to your factory. Moreover, by purchasing exported sodium silicate from RAAG Trading Company, you will also benefit from their special technical and engineering services!


It’s not possible to determine a fixed rate for exported sodium silicate. The price of sodium silicate depends on various factors such as fluctuations in currency exchange rates, the price of oil in the global market, purity percentage, quality level, manufacturer brand, and supplier company. Therefore, we recommend contacting our experts at Raag Trading Company to inquire about the current price, purchase, or export of sodium silicate.
RAAG Trading Company is a reliable choice for purchasing and selling bulk exported sodium silicate by providing high-quality chemicals and detergent raw materials. If you intend to buy or export sodium silicate to other countries, you can contact the sodium silicate sales team at RAAG Trading Company and benefit from their guidance.
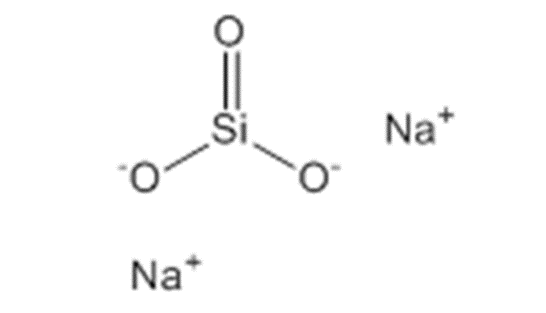
Sodium silicate, also known as water glass, has a general formula of Na2O, xSiO2, where x represents the weight ratio of SiO2 to Na2O, which can range from 1 to 3.2 and is commonly referred to as the “modulus”. In other words, the product of sodium silicate is highly diverse due to the wide range of x values. As the value of x increases, the properties of the sodium silicate product change, and a specific value of x will be suitable for each specific industry. Additionally, liquid sodium silicate products with different purity percentages ranging from 20 to 48 percent are produced depending on the needs of different industries. The weight sum of SiO2 and Na2O determines the purity percentage of the liquid sodium silicate product.
Note: When purchasing sodium silicate for export, be sure to pay attention to its purity percentage and the order of foreign customers.
Sodium silicate is one of the chemical compounds with a wide range of applications in various industries. Those who are involved in the export of sodium silicate are aware of its extensive application. Some of the applications of this product are as follows:
It is used as a buffer material to maintain the alkaline environment of the detergent powders, to stabilize the performance of the detergent material in the powder. In batch formulation, it helps in acid neutralization and also plays a protective role against corrosion in household appliances in detergents, fabric softeners, and bleach. This protective role is achieved by coating the appliances with sodium silicate in combination with the mentioned detergents. Additionally, it has a degree of hardness in detergent powders. In this industry, the modulus of the sodium silicate is 100% in the formulation of detergent powders up to 10% and in bleach up to 0.1%.
Paper Industry:
Sodium silicate is used as an adhesive to give consistency to the pulp in cardboard and paper-making industries. In addition, corrugated sheets are glued together using sodium silicate to form cardboard sheets.
Construction and Tunneling Industry:
It is added to cement to increase the rate of setting. In the tile and ceramic industries, it is used as an anti-cracking adhesive for tiles and ceramics, as well as a viscosity modifier and stabilizer for ceramic slurries. Its concentration in the ceramic slurry formula is up to 5%.
Foundry and Iron Melting Industry:
It is used with special sand for making molds and cores.
Catalyst and Molecular Sieve Industry:
It is used as the main base for the formulation of catalysts and molecular sieves for water and gas purification.
Textile Industry:
Experts use this material to produce flame-resistant clothing.
Crude Oil Refining:
Sodium silicate plays the role of a catalyst in the crude oil refining process.
Non-combustible Industries:
This substance is effective in increasing the stability and non-combustibility of non-combustible industries.
Pipe Insulation:
Industrialists use sodium silicate to increase the heat resistance and material transfer rate in pipes.
Cosmetics and Health Care Industry:
It is used in the production of hair dye, skin care creams, and anti-aging creams.
Water Purification:
Sodium silicate is used in sand filters for water purification.
Water Hardness Reducer:
By weakening the effect of substances such as iron, magnesium, and lead in water, sodium silicate reduces water hardness and prepares it for industrial use.
Soil Stabilizer:
The use of sodium silicate in soil composition improves the flow of electricity through electrodes and stabilizes and improves its quality. Through specific processes and acidification of sodium silicate solutions, silicic acids, which form long polymer chains, can be produced and reached a hydrated material called silica gel. Drying it produces an anhydrous silica gel that is an excellent moisture absorber and has applications in many industries and laboratory purposes.


Sodium silicate is available in the market in both solid and liquid forms, but it is only used through its liquid form. Two methods, wet and dry, are used for the production of sodium silicate. In the wet method, amorphous silica, caustic soda 48%, and water are used. These materials are poured into special reactors and then subjected to the necessary reactions under pressure at a temperature of 180°C with steam at a pressure of 8 bars. Mixing operations are carried out by rotating autoclaves, and after a period of 2 to 4 hours, depending on the desired purity and viscosity, the product is transferred to a special tank or flash tank. After the vapor phase is separated, the remaining solution is filtered, and after the impurities are removed, a transparent sodium silicate solution is obtained, which is ready for delivery to the customer. In this method, the impurity level obtained is up to 10% of the raw materials. The maximum achievable viscosity in the wet method is x = 2.4. To achieve higher x values, the dry or alkali melting furnace method must be used.
2NaOH → Na2O + H2O
Na2O + nSiO2 → Na2O * nSiO2, n = 1/2.4
The dry method requires special furnaces similar to those used in the glass industry. The structure of these furnaces is modern and based on current technology. Various types of refractory bricks are used in the construction of these furnaces. These bricks have different properties, such as corrosion resistance, heat resistance, thermal shock resistance, heat storage and transfer, energy loss insulation, and long life, despite exposure to extremely harsh conditions. The design of the furnaces is such that energy consumption is minimized, and heat recovery from the gases produced by fossil fuel combustion is efficiently and effectively performed with high efficiency.
The raw materials for the export of sodium silicate by the furnace method (alkali melting) include quartz silica powder and heavy sodium carbonate. In the batch plant unit, which includes storage silos, weighing scales, a special solid mixer for mixing silica and carbonate, and a number of vertical and horizontal conveyor belts, the required amount of silica and carbonate powder is weighed depending on the desired viscosity.
After mixing, it is transferred to the storage silo located above the furnace. Then, using the batch charger at a speed proportional to the production plan, the charge is introduced into the furnace. The furnace temperature is adjusted according to the product viscosity and daily draw.
It should be noted that the melting temperature of pure silica is 1700 degrees Celsius, and the melting temperature of pure carbonate is in the range of 750 to 800 degrees Celsius. In the production process of sodium silicate, depending on the amount of these two materials in the batch formulation, the melting temperature is considered in the range of 1150 to 1400 degrees Celsius.
The reaction proceeds according to the following equation:
Na2CO3+XSiO2 → Na2O,XSiO2+CO2
In general, the production process of sodium silicate, particularly through the furnace method, requires high energy consumption. As mentioned, solid sodium silicate must be converted into a solution to be usable for various purposes. The dissolution of silicate rocks, resulting from the furnace, is carried out in the same specialized reactors that produce silicate using the amorphous silica method and soda ash. For this purpose, the silicate rock and water are transferred to an autoclave. If we want to reduce the product’s viscosity, some 48% caustic soda, determined by calculations, is added to the batch. The autoclave door is closed, and it is pressurized with steam. The reaction conditions are 8 bars of steam pressure and a temperature of 180 degrees Celsius.
The formulation of the batch is easily adjusted by stoichiometric calculations, and depending on the customer’s needs (bulk purchase of sodium silicate, export of sodium silicate, or partial purchase), any type of product with the desired viscosity and purity can be produced.
After the mixing time in the autoclave and complete dissolution of the rock, the product is transferred to a flash tank. After the steam is vented, the liquid product, which contains undissolved silicate and impurities present in the raw materials, is transferred to the mixer, where it receives 0.1% perlite as a filter aid.
The turbid product from the mixers is directed towards the filter press with a pressure of at least 5 bars, and after removing impurities, it is directed to storage tanks as a clear solution, ready for presentation to the customer. Please pay attention to the quality of this material for the export of sodium silicate. Raag provides you with the highest quality chemicals.
Unlike the amorphous method, the dissolution of silicate rock in the furnace and the production of a sodium silicate solution from this rock have less impurities, and ultimately, only 4% of the weight of the initial silicate rock is observed as sediment on the filter press.
All production stages, from raw materials to batch preparation in the batch plant, melting and production of silicate rock in furnaces, the dissolution process, including the turbid product, the final product, and also waste monitoring, are under the control of the laboratory quality unit. The final liquid product is stored in steel tanks and transported with 220-liter drums or tankers.
Sodium silicate is supplied in different packaging and weights according to customer requirements. However, this product is sold in 220-liter liquid drums and in 1-ton jumbo bags for solid type, and after placing an order, it will be shipped to the customer’s address. Therefore, for purchasing sodium silicate or inquiring about its price and exporting it, you can place your order online or in-person through the RAAG website.

Although this substance is considered non-toxic, it is recommended to wear gloves, goggles, and special clothing to prevent skin damage while working with it. Also, if you inhale this substance, make sure to go to an open space to avoid any damage to your lungs.
Since sodium silicate is used in various industries, there is a high demand for exporting it. Therefore, you can place your order for sodium silicate online through the Raag website. We will contact you as soon as possible and provide you with the highest quality sodium silicate in standard packaging. Just call the phone numbers listed on this page to get started.